|
When these two conditions are met, the equation is said to be balanced. Also the sum of the charges on one side of the equation must be equal to the sum of the charges on the other side. This means that a chemical equation must have the same number of atoms of each element on both side of the equation. From the balanced chemical equation, use a mole ratio to calculate the number of moles of gold that can be obtained from the. Then calculate the number of moles of Au(CN) 2 present by multiplying the volume of the solution by its concentration. The Law of Conservation of Mass states that mass is neither created nor destroyed in an ordinary chemical reaction. Check the chemical equation to make sure it is balanced as written balance if necessary. If the elements in a chemical formula are properly capitalized, the smart case converter leaves them as you have typed.Ī balanced chemical equation accurately describes the quantities of reactants and products in chemical reactions. 
The equation can be written in lowercase letters.Because the sum of the mass numbers of the reactants must equal the sum of the mass numbers of the products: 25 + 4 A + 1, or A 28 25 + 4 A + 1, or A 28. where A is the mass number and Z is the atomic number of the new nuclide, X. To enter the equation sign, you can use either "=" or "->" or "→" symbols. The nuclear reaction can be written as: 2512Mg +42He 11H +AZX 12 25 Mg + 2 4 He 1 1 H + Z A X. 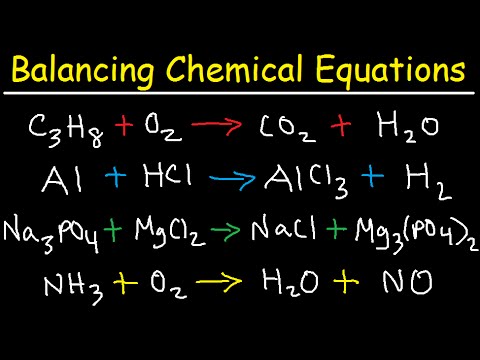
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
